Propane - Prandtl Number vs. Temperature and Pressure
Figures and tables with Prandtl Number of liquid and gaseous propane at varying temperarure and pressure, SI and Imperial units.
The Prandtl Number - Pr - is a dimensionless number approximating the ratio of momentum diffusivity (kinematic viscosity) to thermal diffusivity - and is often used in heat transfer and free and forced convection calculations.
The Prandtl number can for calculations be expressed as
Pr = μ cp / k (1)
where
μ = absolute or dynamic viscosity (kg/(m s)), (lbm/(ft h))
cp = specific heat (J/(kg K)), (Btu/lbmoF)
k = thermal conductivity (W/(m K)), (Btu/(h ft2oF/ft))
Below, Prandtl numbers of propane at varying temperatures and saturation pressure, as well as 1, 10 and 100 bara (14.5, 145 and 1450 psia) are given in figures and tables.
See also other properties of Propane at varying temperature and pressure: Density and specific weight, Dynamic and Kinematic Viscosity, Specific heat (heat capacity), Thermal conductivity and Thermal diffusivity, and Thermophysical properties at standard conditions,
as well as Prandtl number of Air, Ammonia, Carbon dioxide, Methane, Nitrogen and Water.
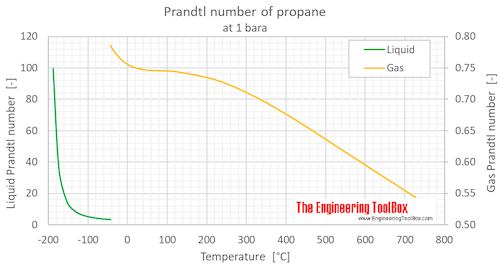
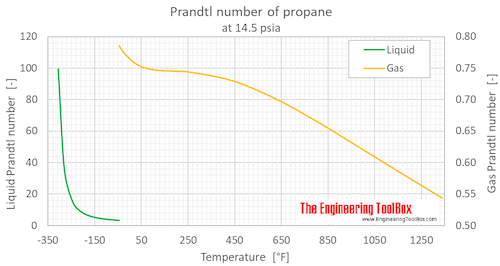
Prandtl number of propane at atmospheric pressure, varying temperature given as °C or °F:
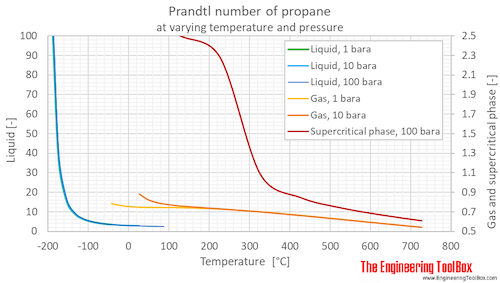
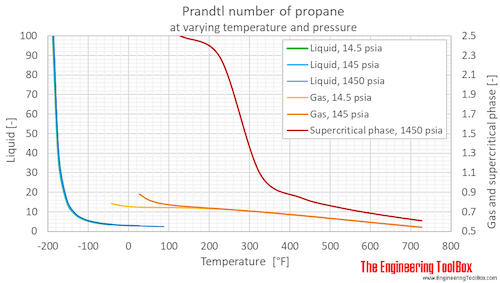
Prandtl number of propane at 1, 10 and 100 bara (14.5, 145 and 1450 psia), varying temperature given as °C or °F:
Prandtl number of propane at atmospheric pressure, temperature given as K, °C or °F:
| State | Temperature | Prandtl number | Temperature | Prandtl number | |||
|---|---|---|---|---|---|---|---|
| (K) | (°C) | (-) | (K) | (°F) | (-) | ||
| Liquid | 123 | -150 | 13.7 | 116 | -250 | 17.0 | |
| 148 | -125 | 7.59 | 144 | -200 | 8.15 | ||
| 173 | -100 | 5.32 | 172 | -150 | 5.39 | ||
| 198 | -75 | 4.20 | 200 | -100 | 4.15 | ||
| 223 | -50 | 3.57 | 228 | -50 | 3.48 | ||
| 231 | -42 | 3.43 | 231 | -44 | 3.43 | ||
| Gas | 231 | -42 | 0.784 | 231 | -44 | 0.784 | |
| 248 | -25 | 0.770 | 244 | -20 | 0.773 | ||
| 263 | -10 | 0.761 | 255 | 0 | 0.766 | ||
| 268 | -5 | 0.759 | 266 | 20 | 0.760 | ||
| 273 | 0 | 0.757 | 272 | 30 | 0.757 | ||
| 278 | 5 | 0.755 | 278 | 40 | 0.755 | ||
| 283 | 10 | 0.753 | 283 | 50 | 0.753 | ||
| 293 | 20 | 0.751 | 294 | 70 | 0.750 | ||
| 298 | 25 | 0.750 | 300 | 80 | 0.749 | ||
| 303 | 30 | 0.749 | 305 | 90 | 0.748 | ||
| 323 | 50 | 0.746 | 311 | 100 | 0.747 | ||
| 348 | 75 | 0.745 | 339 | 150 | 0.745 | ||
| 373 | 100 | 0.744 | 366 | 200 | 0.744 | ||
| 398 | 125 | 0.744 | 394 | 250 | 0.744 | ||
| 423 | 150 | 0.743 | 422 | 300 | 0.743 | ||
| 448 | 175 | 0.740 | 478 | 400 | 0.736 | ||
| 473 | 200 | 0.737 | 533 | 500 | 0.723 | ||
| 523 | 250 | 0.726 | 589 | 600 | 0.706 | ||
| 573 | 300 | 0.711 | 644 | 700 | 0.686 | ||
| 623 | 350 | 0.694 | 700 | 800 | 0.665 | ||
| 673 | 400 | 0.675 | 755 | 900 | 0.644 | ||
| 773 | 500 | 0.637 | 811 | 1000 | 0.622 | ||
Prandtl number of propane at given temperatures and saturation pressure as well as 1, 10 and 100 bara (14.5, 145 and 1450 psia):
| State | Temperature | Pressure | Prandtl number | |||
|---|---|---|---|---|---|---|
| (K) | (°C) | (°F) | (bara) | (psia) | (-) | |
| Liquid at
equilibrium |
100 | -173 | -280 | 2.5E-07 | 3.7E-06 | 35.37 |
| 130 | -143 | -226 | 0.000176 | 0.00255 | 11.19 | |
| 160 | -113 | -172 | 0.00850 | 0.1233 | 6.270 | |
| 190 | -83.2 | -118 | 0.1055 | 1.530 | 4.495 | |
| 220 | -53.2 | -63.7 | 0.6058 | 8.787 | 3.643 | |
| 250 | -23.2 | -9.7 | 2.180 | 31.62 | 3.179 | |
| 280 | 6.9 | 44.3 | 5.817 | 84.37 | 2.920 | |
| 310 | 36.9 | 98.3 | 12.73 | 184.6 | 2.790 | |
| 320 | 46.9 | 116 | 15.99 | 231.9 | 2.770 | |
| 340 | 66.9 | 152 | 24.32 | 352.7 | 2.848 | |
| 355 | 81.9 | 179 | 32.43 | 470.4 | 3.259 | |
| 360 | 86.9 | 188 | 35.55 | 515.6 | 3.548 | |
|
Gas at equilibrium |
100 | -173 | -280 | 2.5E-07 | 3.7E-06 | 1.154 |
| 130 | -143 | -226 | 0.000176 | 0.00255 | 0.9481 | |
| 160 | -113 | -172 | 0.00850 | 0.1233 | 0.8499 | |
| 190 | -83.2 | -118 | 0.1055 | 1.530 | 0.8002 | |
| 220 | -53.2 | -63.7 | 0.6058 | 8.787 | 0.7840 | |
| 250 | -23.2 | -9.7 | 2.180 | 31.62 | 0.7956 | |
| 280 | 6.9 | 44.3 | 5.817 | 84.37 | 0.8357 | |
| 310 | 36.9 | 98.3 | 12.73 | 184.6 | 0.9249 | |
| 320 | 46.9 | 116 | 15.99 | 231.9 | 0.9789 | |
| 340 | 66.9 | 152 | 24.32 | 352.7 | 1.188 | |
| 355 | 81.9 | 179 | 32.43 | 470.4 | 1.732 | |
| 360 | 86.9 | 188 | 35.55 | 515.6 | 2.300 | |
| Liquid | 85.53 | -187.62 | -305.72 | 1 | 14.5 | 99.44 |
| 100 | -173.15 | -279.67 | 1 | 14.5 | 35.88 | |
| 120 | -153.15 | -243.67 | 1 | 14.5 | 15.11 | |
| 140 | -133.15 | -207.67 | 1 | 14.5 | 8.891 | |
| 160 | -113.15 | -171.67 | 1 | 14.5 | 6.290 | |
| 180 | -93.15 | -135.67 | 1 | 14.5 | 4.942 | |
| 200 | -73.15 | -99.67 | 1 | 14.5 | 4.145 | |
| 220 | -53.15 | -63.67 | 1 | 14.5 | 3.633 | |
| 230.74 | -42.41 | -44.34 | 1 | 14.5 | 3.433 | |
| Gas | 230.74 | -42.41 | -44.34 | 1 | 14.5 | 0.7857 |
| 240 | -33.2 | -27.67 | 1 | 14.5 | 0.7760 | |
| 260 | -13.2 | 8.33 | 1 | 14.5 | 0.7621 | |
| 280 | 6.9 | 44.33 | 1 | 14.5 | 0.7534 | |
| 300 | 26.9 | 80.33 | 1 | 14.5 | 0.7492 | |
| 320 | 46.9 | 116 | 1 | 14.5 | 0.7467 | |
| 340 | 66.9 | 152 | 1 | 14.5 | 0.7458 | |
| 360 | 86.9 | 188 | 1 | 14.5 | 0.7453 | |
| 400 | 127 | 260 | 1 | 14.5 | 0.7437 | |
| 500 | 227 | 440 | 1 | 14.5 | 0.7301 | |
| 600 | 327 | 620 | 1 | 14.5 | 0.7024 | |
| 700 | 427 | 800 | 1 | 14.5 | 0.6658 | |
| 800 | 527 | 980 | 1 | 14.5 | 0.6252 | |
| 900 | 627 | 1160 | 1 | 14.5 | 0.5840 | |
| 1000 | 727 | 1340 | 1 | 14.5 | 0.5434 | |
| Liquid | 85.62 | -187.53 | -305.55 | 10 | 145 | 99.81 |
| 100 | -173 | -280 | 10 | 145 | 36.19 | |
| 120 | -153 | -244 | 10 | 145 | 15.21 | |
| 140 | -133 | -208 | 10 | 145 | 8.941 | |
| 160 | -113 | -172 | 10 | 145 | 6.318 | |
| 180 | -93.2 | -136 | 10 | 145 | 4.958 | |
| 200 | -73.2 | -99.7 | 10 | 145 | 4.156 | |
| 220 | -53.2 | -63.7 | 10 | 145 | 3.641 | |
| 240 | -33.2 | -27.7 | 10 | 145 | 3.300 | |
| 260 | -13.2 | 8.3 | 10 | 145 | 3.064 | |
| 280 | 6.9 | 44.3 | 10 | 145 | 2.909 | |
| 300.09 | 26.94 | 80.49 | 10 | 145 | 2.811 | |
| Gas | 300.09 | 26.94 | 80.49 | 10 | 145 | 0.8851 |
| 320 | 46.9 | 116 | 10 | 145 | 0.8299 | |
| 340 | 66.9 | 152 | 10 | 145 | 0.8001 | |
| 360 | 86.9 | 188 | 10 | 145 | 0.7824 | |
| 400 | 127 | 260 | 10 | 145 | 0.7619 | |
| 500 | 227 | 440 | 10 | 145 | 0.7321 | |
| 600 | 327 | 620 | 10 | 145 | 0.6999 | |
| 700 | 427 | 800 | 10 | 145 | 0.6626 | |
| 800 | 527 | 980 | 10 | 145 | 0.6218 | |
| 900 | 627 | 1160 | 10 | 145 | 0.5810 | |
| 1000 | 727 | 1340 | 10 | 145 | 0.5407 | |
| Liquid | 100 | -173 | -280 | 50 | 725 | 37.10 |
| 200 | -73.2 | -99.7 | 50 | 725 | 4.216 | |
| 300 | 26.9 | 80.3 | 50 | 725 | 2.802 | |
| Supercritical phase |
400 | 127 | 260 | 50 | 725 | 1.305 |
| 500 | 227 | 440 | 50 | 725 | 0.8190 | |
| Liquid | 86.45 | -187 | -304 | 100 | 1450 | 103.18 |
| 100 | -173 | -280 | 100 | 1450 | 39.46 | |
| 120 | -153 | -244 | 100 | 1450 | 16.28 | |
| 140 | -133 | -208 | 100 | 1450 | 9.433 | |
| 160 | -113 | -172 | 100 | 1450 | 6.596 | |
| 180 | -93.2 | -136 | 100 | 1450 | 5.139 | |
| 200 | -73.2 | -99.7 | 100 | 1450 | 4.282 | |
| 220 | -53.2 | -63.7 | 100 | 1450 | 3.735 | |
| 240 | -33.2 | -27.7 | 100 | 1450 | 3.367 | |
| 260 | -13.2 | 8.3 | 100 | 1450 | 3.112 | |
| 280 | 6.9 | 44.3 | 100 | 1450 | 2.934 | |
| 300 | 26.9 | 80.3 | 100 | 1450 | 2.798 | |
| 320 | 46.9 | 116 | 100 | 1450 | 2.691 | |
| 340 | 66.9 | 152 | 100 | 1450 | 2.597 | |
| 360 | 86.9 | 188 | 100 | 1450 | 2.504 | |
| Supercritical phase |
400 | 127 | 260 | 100 | 1450 | 2.282 |
| 500 | 227 | 440 | 100 | 1450 | 1.085 | |
| 600 | 327 | 620 | 100 | 1450 | 0.8466 | |
| 700 | 427 | 800 | 100 | 1450 | 0.7417 | |
| 800 | 527 | 980 | 100 | 1450 | 0.6694 | |
| 900 | 627 | 1160 | 100 | 1450 | 0.6098 | |
| 1000 | 727 | 1340 | 100 | 1450 | 0.5586 | |



