Ethanol - Specific Heat vs. Temperature and Pressure
Online calculators, figures and tables showing specific heat , Cp and Cv, of gasous and liquid ethanol at temperatures ranging from -25 to 325 °C (-10 to 620 °F) at atmospheric and higher pressure - Imperial and SI Units.
Specific heat (C) is the amount of heat required to change the temperature of a mass unit of a substance by one degree.
- Isobaric specific heat (Cp) is used for ethanol in a constant pressure (ΔP = 0) system.
- I sochoric specific heat (Cv) is used for ethanol in a constant-volume , (= isovolumetric or isometric ) closed system.
At ambient pressure and temperature the isobaric specific heat, CP, of liquid ethanol is 2.57 (kJ/kg K) or 0.614 (Btu/lb °F) = (cal/g K), while the isochoric specific heat, CV, is 2.18 (kJ/kg K) or 0.520 (Btu/lb °F) = (cal/g K).
However, the specific heat - CP and CV - will vary with temperature. This may influence on the accuracy of ethanol conditioning and industrial ethanol handling process calculations. When calculating mass and volume flow of ethanol in heated or cooled systems with high accuracy - the specific heat (= heat capacity) should be corrected according values in the figures and table below, or found by use of the calculator.
- Specific heat unit conversion
Online Liquid Ethanol Specific Heat Calculator
The calculator below can be used to estimate the specific heat of liquid ethanol at constant volum(CV) or constant pressure(CP) and at given temperature. For practical purpose the specific heat of liquid ethanol is constant with varying pressure up to critical point (241°C, 61.5 bara / 465°F, 892 psia).
The output specific heat is given as kJ/(kmol*K), kJ/(kg*K), kWh/(kg*K), kcal/(kg*K), Btu(IT)/(mol*°R) and Btu(IT)/(lbm *°R)
See also other properties of Ethanol at varying temperature and pressure : Density and specific weight and Dynamic and kinematic viscosity, and Thermophysical properties at standard conditions,
as well as Specific heat (Heat capacity) of Air - at Constant Pressure and Varying Temperature, Air - at Constant Temperature and Varying Pressure, Ammonia, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethylene, Hydrogen, Methane, Methanol, Nitrogen, Oxygen, Propane and Water .
Isobaric specific heat, CP, of ethanol at given temperatures and pressure s:
For full table with Imperial units - rotate the screen!
| State | Temperature | Pressure | Specific heat at constant pressure, CP (isobaric) | Cp/Cv | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (K) | (°C) | (°F) | (MPa) | (bara) | (psia) | (kJ/mol K) | (kJ/kg K) | (kWh/(kg K)) | (kcal(IT)/(kg K)), (Btu(IT)/lb °F) | (Btu(IT)/(mol*°R)) | (-) | |
| Liquid | 250 | -23.2 | -9.7 | 0.000270 | 0.00270 | 0.0392 | 0.09361 | 2.032 | 0.000564 | 0.4853 | 0.04929 | 1.22 |
| 280 | 6.9 | 44.3 | 0.00258 | 0.0258 | 0.375 | 0.1083 | 2.351 | 0.000653 | 0.5614 | 0.05702 | 1.18 | |
| 298.2 | 25.0 | 77.0 | 0.0080 | 0.080 | 1.16 | 0.1184 | 2.570 | 0.000714 | 0.6138 | 0.06234 | 1.18 | |
| 310 | 36.9 | 98.3 | 0.0153 | 0.153 | 2.22 | 0.1252 | 2.719 | 0.000755 | 0.6493 | 0.06595 | 1.17 | |
| 340 | 66.9 | 152 | 0.0635 | 0.635 | 9.22 | 0.1412 | 3.064 | 0.000851 | 0.7318 | 0.07432 | 1.19 | |
| 370 | 96.9 | 206 | 0.2021 | 2.021 | 29.30 | 0.1554 | 3.374 | 0.000937 | 0.8058 | 0.08184 | 1.22 | |
| 400 | 127 | 260 | 0.5245 | 5.245 | 76.07 | 0.1688 | 3.665 | 0.00102 | 0.8753 | 0.08890 | 1.26 | |
| 423.9 | 151 | 303 | 1.000 | 10.00 | 145.0 | 0.1802 | 3.910 | 0.00109 | 0.9340 | 0.09486 | 1.31 | |
| 430 | 157 | 314 | 1.165 | 11.65 | 169.0 | 0.1834 | 3.981 | 0.00111 | 0.9509 | 0.09658 | 1.33 | |
| 460 | 187 | 368 | 2.292 | 22.92 | 332.4 | 0.2050 | 4.451 | 0.00124 | 1.063 | 0.1080 | 1.46 | |
| 490 | 217 | 422 | 4.095 | 40.95 | 594.0 | 0.2651 | 5.754 | 0.00160 | 1.374 | 0.1396 | 1.86 | |
| 501.4 | 228 | 443 | 5.000 | 50.00 | 725.2 | 0.3515 | 7.630 | 0.00212 | 1.822 | 0.1851 | 2.45 | |
| Gas | 250 | -23.2 | -9.7 | 0.000270 | 0.00270 | 0.0392 | 0.06722 | 1.459 | 0.000405 | 0.3485 | 0.03539 | 1.14 |
| 280 | 6.9 | 44.3 | 0.00258 | 0.0258 | 0.375 | 0.07115 | 1.544 | 0.000429 | 0.3689 | 0.03746 | 1.13 | |
| 298.2 | 25.0 | 77.0 | 0.0080 | 0.0800 | 1.16 | 0.07380 | 1.602 | 0.000445 | 0.3826 | 0.03886 | 1.13 | |
| 310 | 36.9 | 98.3 | 0.0153 | 0.153 | 2.22 | 0.07546 | 1.638 | 0.000455 | 0.3912 | 0.03974 | 1.13 | |
| 340 | 66.9 | 152 | 0.0635 | 0.635 | 9.22 | 0.08074 | 1.753 | 0.000487 | 0.4186 | 0.04251 | 1.13 | |
| 370 | 96.9 | 206 | 0.2021 | 2.021 | 29.30 | 0.08806 | 1.911 | 0.000531 | 0.4565 | 0.04637 | 1.15 | |
| 400 | 127 | 260 | 0.5245 | 5.245 | 76.07 | 0.09894 | 2.148 | 0.000597 | 0.5129 | 0.05210 | 1.18 | |
| 423.9 | 151 | 303 | 1.000 | 10.00 | 145.0 | 0.1118 | 2.428 | 0.000674 | 0.5798 | 0.05889 | 1.24 | |
| 430 | 157 | 314 | 1.165 | 11.65 | 169.0 | 0.1161 | 2.520 | 0.000700 | 0.6019 | 0.06113 | 1.26 | |
| 460 | 187 | 368 | 2.292 | 22.92 | 332.4 | 0.1473 | 3.197 | 0.000888 | 0.7635 | 0.07755 | 1.43 | |
| 490 | 217 | 422 | 4.095 | 40.95 | 594.0 | 0.2320 | 5.036 | 0.00140 | 1.203 | 0.1222 | 1.98 | |
| 501.4 | 228 | 443 | 5.000 | 50.00 | 725.2 | 0.3410 | 7.402 | 0.00206 | 1.768 | 0.1796 | 2.75 | |
| Liquid | 300 | 26.9 | 80.3 | 0.1 | 1 | 14.5 | 0.1196 | 2.597 | 0.000721 | 0.6202 | 0.06299 | 1.17 |
| 351.1 | 77.9 | 172 | 0.1 | 1 | 14.5 | 0.1466 | 3.182 | 0.000884 | 0.7599 | 0.07718 | 1.20 | |
| Gas | 351.1 | 77.9 | 172 | 0.1 | 1 | 14.5 | 0.08313 | 1.804 | 0.000501 | 0.4310 | 0.04377 | 1.14 |
| 400 | 127 | 260 | 0.1 | 1 | 14.5 | 0.09000 | 1.954 | 0.000543 | 0.4666 | 0.04739 | 1.12 | |
| 500 | 227 | 440 | 0.1 | 1 | 14.5 | 0.1016 | 2.206 | 0.000613 | 0.5268 | 0.05351 | 1.09 | |
| 600 | 327 | 620 | 0.1 | 1 | 14.5 | 0.1125 | 2.442 | 0.000678 | 0.5834 | 0.05925 | 1.08 | |
| Liquid | 300 | 26.9 | 80.3 | 1 | 10 | 145 | 0.1195 | 2.595 | 0.000721 | 0.6198 | 0.06294 | 1.17 |
| 400 | 127 | 260 | 1 | 10 | 145 | 0.1686 | 3.659 | 0.001016 | 0.8740 | 0.08876 | 1.26 | |
| 423.9 | 151 | 303 | 1 | 10 | 145 | 0.1802 | 3.910 | 0.001086 | 0.9340 | 0.09486 | 1.31 | |
| Gas | 423.9 | 151 | 303 | 1 | 10 | 145 | 0.1118 | 2.428 | 0.000674 | 0.5798 | 0.05889 | 1.24 |
| 500 | 227 | 440 | 1 | 10 | 145 | 0.1101 | 2.389 | 0.000664 | 0.5707 | 0.05796 | 1.14 | |
| 600 | 327 | 620 | 1 | 10 | 145 | 0.1161 | 2.519 | 0.000700 | 0.6017 | 0.06111 | 1.10 | |
| Liquid | 300 | 26.9 | 80.3 | 5 | 50 | 725 | 0.1192 | 2.588 | 0.000719 | 0.6181 | 0.06278 | 1.17 |
| 400 | 127 | 260 | 5 | 50 | 725 | 0.1666 | 3.616 | 0.00100 | 0.8636 | 0.08771 | 1.25 | |
| 501.4 | 228 | 443 | 5 | 50 | 725 | 0.3515 | 7.630 | 0.00212 | 1.822 | 0.1851 | 2.45 | |
| Gas | 501.4 | 228 | 443 | 5 | 50 | 725 | 0.3410 | 7.402 | 0.00206 | 1.768 | 0.1796 | 2.75 |
| 600 | 327 | 620 | 5 | 50 | 725 | 0.1366 | 2.965 | 0.000824 | 0.7082 | 0.07192 | 1.20 | |
| Liquid | 300 | 26.9 | 80.3 | 10 | 100 | 1450 | 0.1189 | 2.580 | 0.000717 | 0.6162 | 0.06258 | 1.17 |
| Supercritical phase |
400 | 127 | 260 | 10 | 100 | 1450 | 0.1646 | 3.572 | 0.000992 | 0.8532 | 0.08665 | 1.24 |
| 500 | 227 | 440 | 10 | 100 | 1450 | 0.2220 | 4.820 | 0.00134 | 1.151 | 0.1169 | 1.58 | |
| 600 | 327 | 620 | 10 | 100 | 1450 | 0.1874 | 4.069 | 0.00113 | 0.9718 | 0.09870 | 1.49 | |
| Liquid | 300 | 26.9 | 80.3 | 100 | 1000 | 14500 | 0.1157 | 2.512 | 0.000698 | 0.5999 | 0.06093 | 1.14 |
| Supercritical phase |
400 | 127 | 260 | 100 | 1000 | 14500 | 0.1508 | 3.274 | 0.000909 | 0.7819 | 0.07941 | 1.17 |
| 500 | 227 | 440 | 100 | 1000 | 14500 | 0.1643 | 3.567 | 0.000991 | 0.8520 | 0.08653 | 1.24 | |
| 600 | 327 | 620 | 100 | 1000 | 14500 | 0.1655 | 3.593 | 0.000998 | 0.8582 | 0.08716 | 1.32 | |
| Supercritical phase |
300 | 26.9 | 80.3 | 200 | 2000 | 29000 | 0.1150 | 2.495 | 0.000693 | 0.5960 | 0.06053 | 1.13 |
| 400 | 127 | 260 | 200 | 2000 | 29000 | 0.1454 | 3.156 | 0.000877 | 0.7538 | 0.07656 | 1.15 | |
| 500 | 227 | 440 | 200 | 2000 | 29000 | 0.1562 | 3.391 | 0.000942 | 0.8100 | 0.08226 | 1.21 | |
| 600 | 327 | 620 | 200 | 2000 | 29000 | 0.1557 | 3.379 | 0.000939 | 0.8070 | 0.08196 | 1.29 | |
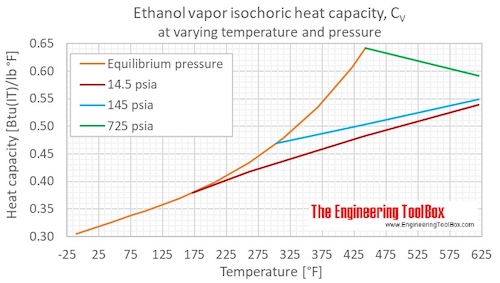
Isochoric specific heat, CV, of ethanol at given temperatures and pressure s:
For full table with Imperial units - rotate the screen!
| State | Temperature | Pressure | Specific heat at constant volume, CV (isochoric) | Cp/Cv | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (K) | (°C) | (°F) | (MPa) | (bara) | (psia) | (kJ/mol K) | (kJ/kg K) | (kWh/(kg K)) | (kcal(IT)/(kg K)), (Btu(IT)/lb °F) | (Btu(IT)/(mol*°R)) | (-) | |
|
Liquid at |
250 | -23.2 | -9.7 | 0.000270 | 0.00270 | 0.0392 | 0.07666 | 1.664 | 0.000462 | 0.3974 | 0.04036 | 1.22 |
| 280 | 6.9 | 44.3 | 0.00258 | 0.0258 | 0.375 | 0.09165 | 1.989 | 0.000553 | 0.4752 | 0.04826 | 1.18 | |
| 298.2 | 25.0 | 77.0 | 0.0080 | 0.080 | 1.16 | 0.1003 | 2.178 | 0.000605 | 0.5202 | 0.05283 | 1.18 | |
| 310 | 36.9 | 98.3 | 0.0153 | 0.153 | 2.22 | 0.1067 | 2.316 | 0.000643 | 0.5532 | 0.05618 | 1.17 | |
| 340 | 66.9 | 152 | 0.0635 | 0.635 | 9.22 | 0.1189 | 2.582 | 0.000717 | 0.6166 | 0.06262 | 1.19 | |
| 370 | 96.9 | 206 | 0.2021 | 2.021 | 29.30 | 0.1279 | 2.777 | 0.000771 | 0.6632 | 0.06736 | 1.22 | |
| 400 | 127 | 260 | 0.5245 | 5.245 | 76.07 | 0.1341 | 2.910 | 0.000808 | 0.6950 | 0.07059 | 1.26 | |
| 423.9 | 151 | 303 | 1.000 | 10.00 | 145.0 | 0.1373 | 2.981 | 0.000828 | 0.7119 | 0.07231 | 1.31 | |
| 430 | 157 | 314 | 1.165 | 11.65 | 169.0 | 0.1380 | 2.995 | 0.000832 | 0.7154 | 0.07265 | 1.33 | |
| 460 | 187 | 368 | 2.292 | 22.92 | 332.4 | 0.1404 | 3.048 | 0.000847 | 0.7280 | 0.07393 | 1.46 | |
| 490 | 217 | 422 | 4.095 | 40.95 | 594.0 | 0.1423 | 3.090 | 0.000858 | 0.7380 | 0.07495 | 1.86 | |
| 501.4 | 228 | 443 | 5.000 | 50.00 | 725.2 | 0.1434 | 3.113 | 0.000865 | 0.7435 | 0.07551 | 2.45 | |
| Gas at equilibrium |
250 | -23.2 | -9.7 | 0.000270 | 0.00270 | 0.0392 | 0.05889 | 1.278 | 0.000355 | 0.3053 | 0.03101 | 1.14 |
| 280 | 6.9 | 44.3 | 0.00258 | 0.0258 | 0.375 | 0.06275 | 1.362 | 0.000378 | 0.3253 | 0.03304 | 1.13 | |
| 298.2 | 25.0 | 77.0 | 0.0080 | 0.080 | 1.16 | 0.06531 | 1.418 | 0.000394 | 0.3386 | 0.03439 | 1.13 | |
| 310 | 36.9 | 98.3 | 0.0153 | 0.153 | 2.22 | 0.06682 | 1.450 | 0.000403 | 0.3464 | 0.03518 | 1.13 | |
| 340 | 66.9 | 152 | 0.0635 | 0.635 | 9.22 | 0.07134 | 1.548 | 0.000430 | 0.3698 | 0.03756 | 1.13 | |
| 370 | 96.9 | 206 | 0.2021 | 2.021 | 29.30 | 0.07684 | 1.668 | 0.000463 | 0.3984 | 0.04046 | 1.15 | |
| 400 | 127 | 260 | 0.5245 | 5.245 | 76.07 | 0.08377 | 1.818 | 0.000505 | 0.4343 | 0.04411 | 1.18 | |
| 423.9 | 151 | 303 | 1.000 | 10.00 | 145.0 | 0.09052 | 1.965 | 0.000546 | 0.4693 | 0.04766 | 1.24 | |
| 430 | 157 | 314 | 1.165 | 11.65 | 169.0 | 0.09245 | 2.007 | 0.000557 | 0.4793 | 0.04868 | 1.26 | |
| 460 | 187 | 368 | 2.292 | 22.92 | 332.4 | 0.1032 | 2.241 | 0.000622 | 0.5352 | 0.05436 | 1.43 | |
| 490 | 217 | 422 | 4.095 | 40.95 | 594.0 | 0.1171 | 2.542 | 0.000706 | 0.6071 | 0.06165 | 1.98 | |
| 501.4 | 228 | 443 | 5.000 | 50.00 | 725.2 | 0.1239 | 2.689 | 0.000747 | 0.6423 | 0.06524 | 2.75 | |
| Liquid | 300 | 26.9 | 80.3 | 0.1 | 1 | 14.5 | 0.1019 | 2.213 | 0.000615 | 0.5285 | 0.05367 | 1.17 |
| 351.1 | 77.9 | 172 | 0.1 | 1 | 14.5 | 0.1226 | 2.661 | 0.000739 | 0.6357 | 0.06456 | 1.20 | |
| Gas | 351.1 | 77.9 | 172 | 0.1 | 1 | 14.5 | 0.07322 | 1.589 | 0.000441 | 0.3796 | 0.03856 | 1.14 |
| 400 | 127 | 260 | 0.1 | 1 | 14.5 | 0.08064 | 1.750 | 0.000486 | 0.4181 | 0.04246 | 1.12 | |
| 500 | 227 | 440 | 0.1 | 1 | 14.5 | 0.09291 | 2.017 | 0.000560 | 0.4817 | 0.04892 | 1.09 | |
| 600 | 327 | 620 | 0.1 | 1 | 14.5 | 0.10403 | 2.258 | 0.000627 | 0.5393 | 0.05478 | 1.08 | |
| Liquid | 300 | 26.9 | 80.3 | 1 | 10 | 145 | 0.1019 | 2.212 | 0.000614 | 0.5284 | 0.05366 | 1.17 |
| 400 | 127 | 260 | 1 | 10 | 145 | 0.1340 | 2.909 | 0.000808 | 0.6947 | 0.07056 | 1.26 | |
| 423.9 | 151 | 303 | 1 | 10 | 145 | 0.1373 | 2.981 | 0.000828 | 0.7119 | 0.07231 | 1.31 | |
| Gas | 423.9 | 151 | 303 | 1 | 10 | 145 | 0.09052 | 1.965 | 0.000546 | 0.4693 | 0.04766 | 1.24 |
| 500 | 227 | 440 | 1 | 10 | 145 | 0.09695 | 2.105 | 0.000585 | 0.5027 | 0.05105 | 1.14 | |
| 600 | 327 | 620 | 1 | 10 | 145 | 0.10581 | 2.297 | 0.000638 | 0.5486 | 0.05572 | 1.10 | |
| Liquid | 300 | 26.9 | 80.3 | 5 | 50 | 725 | 0.1019 | 2.211 | 0.000614 | 0.5280 | 0.05363 | 1.17 |
| 400 | 127 | 260 | 5 | 50 | 725 | 0.1336 | 2.900 | 0.000806 | 0.6926 | 0.07034 | 1.25 | |
| 501.4 | 228 | 443 | 5 | 50 | 725 | 0.1434 | 3.113 | 0.000865 | 0.7435 | 0.07551 | 2.45 | |
| Gas | 501.4 | 228 | 443 | 5 | 50 | 725 | 0.1239 | 2.689 | 0.000747 | 0.6423 | 0.06524 | 2.75 |
| 600 | 327 | 620 | 5 | 50 | 725 | 0.1142 | 2.479 | 0.000689 | 0.5920 | 0.06013 | 1.20 | |
| Liquid | 300 | 26.9 | 80.3 | 10 | 100 | 1450 | 0.1018 | 2.210 | 0.000614 | 0.5277 | 0.05360 | 1.17 |
| Supercritical phase |
400 | 127 | 260 | 10 | 100 | 1450 | 0.1331 | 2.890 | 0.000803 | 0.6902 | 0.07010 | 1.24 |
| 500 | 227 | 440 | 10 | 100 | 1450 | 0.1403 | 3.046 | 0.000846 | 0.7274 | 0.07388 | 1.58 | |
| 600 | 327 | 620 | 10 | 100 | 1450 | 0.1260 | 2.735 | 0.000760 | 0.6532 | 0.06634 | 1.49 | |
| Liquid | 300 | 26.9 | 80.3 | 100 | 1000 | 14500 | 0.1015 | 2.203 | 0.000612 | 0.5262 | 0.05344 | 1.14 |
| Supercritical phase |
400 | 127 | 260 | 100 | 1000 | 14500 | 0.1290 | 2.800 | 0.000778 | 0.6689 | 0.06793 | 1.17 |
| 500 | 227 | 440 | 100 | 1000 | 14500 | 0.1322 | 2.870 | 0.000797 | 0.6854 | 0.06962 | 1.24 | |
| 600 | 327 | 620 | 100 | 1000 | 14500 | 0.1258 | 2.730 | 0.000758 | 0.6520 | 0.06621 | 1.32 | |
| Supercritical phase |
300 | 26.9 | 80.3 | 200 | 2000 | 29000 | 0.1020 | 2.213 | 0.000615 | 0.5286 | 0.05369 | 1.13 |
| 400 | 127 | 260 | 200 | 2000 | 29000 | 0.1268 | 2.752 | 0.000764 | 0.6573 | 0.06676 | 1.15 | |
| 500 | 227 | 440 | 200 | 2000 | 29000 | 0.1287 | 2.793 | 0.000776 | 0.6671 | 0.06776 | 1.21 | |
| 600 | 327 | 620 | 200 | 2000 | 29000 | 0.1207 | 2.619 | 0.000728 | 0.6256 | 0.06353 | 1.29 | |
british thermal unit(International table) = (Btu(IT)), degree celcius = (°C), degree fahrenheit = (°F), degree kelvin = (K), degree rankin = (°R), joule = (J), kilocalorie(International table) = (kcal(IT)), kilogram = (kg), kilojoule = (kJ), kilowatthour = (kWh), mole = (mol), pound =(lb)
K in the units can be replaced by °C, and vise versa. °R in the units can be replaced by °F, and vise versa.
- 1 Btu/(lb °F) = 1 Btu/(lb °R) = 1 kcal(IT)/(kg °C) = 1 kcal(IT)/(kg K) = 4186.8 J/(kg K) = 0.81647 kcal(IT)/(lb °F) = 1.163×10-3 kWh/(kg K)
- 1 J/(kg K) = 1 J/(kg °C) = 2.3885×10-4 kcal(IT)/(kg oC) = 2.3885×10-4 Btu/(lb °F) = 1.9501×10-4 kcal(IT)/(lb °F)
- 1 kcal(IT)/(kg °C) = 1 Btu/(lb °F) = 4186.8 J/(kg K) = 0.81647 kcal(IT)/(lb °F) = 1.163×10-3 kWh/(kg K)
- 1 kcal(IT)/(lb °F) = 1.2248 Btu/(lb °F) = 1.2248 kcal(IT)/(kg °C) = 5127.9 J/(kg K)
- 1 kJ/kgK = 1 kJ/(kg °C) = 1000 J/(kg K) = 1000 J/(kg °C) = 0.23885 kcal(IT)/(kg °C) = 0.23885 Btu/(lb °F) = 0.19501 kcal(IT)/(lb °F) = 2.7778×10-4 kWh/(kg K)
- 1 kWh/(kg K) = 0.85985 kcal(IT)/(kg °C) = 0.85985 Btu/(lb °F) = 3.6 kJ/kgK
- 1 mol of air = 28.96546 g