Densities of Aqueous Solutions of Inorganic Potassium Salts
Changes in density of aqueous solutions with changes in concentration at 20°C. Density of potassium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.
Be aware of the concentration units in the figures:
wt%: Mass of solute/total mass of solution ×100%
mol/kg: Molality = moles of solute/kg of water
mol/liter: Molarity = moles of solute/liter of solution
Values are tabulated below the figures.
See also density of aqueous solutions of inorganic chlorides, inorganic sodium salts, some other inorganic substances, organic acids and organic substances as sugars and alcohols.

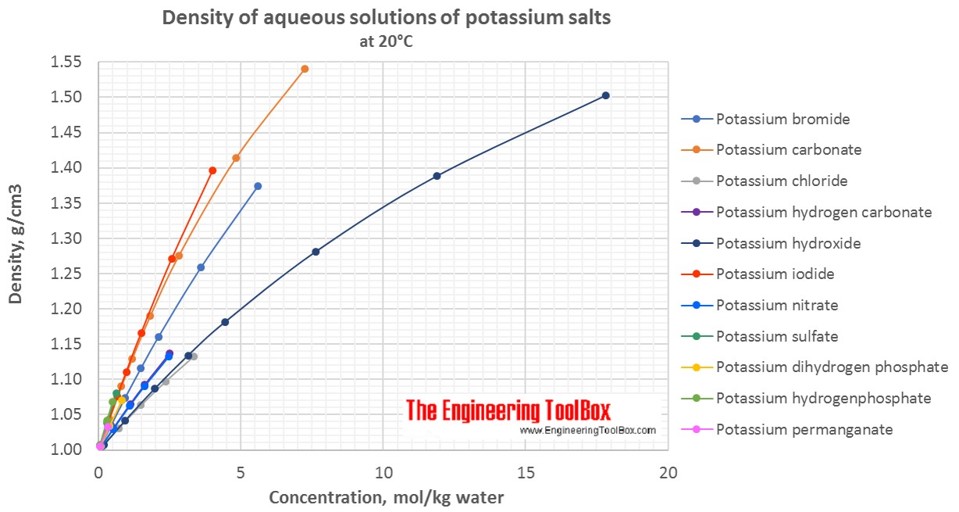


Density of aqueous solutions at 20°C, given as g/cm3:
For full table with Potassium Hydrogen Carbonate, Potassium Hydrogen Phosphate, Potassium Hydroxide, Potassium Iodide, Potassium Nitrate, Potassium Permanganate and Potassium Sulfate - rotate the screen!
| Mass% | Potassium bromide | Potassium carbonate | Potassium chloride | Potassium dihydrogen phosphate | Potassium hydrogen carbonate | Potassium hydrogen phosphate | Potassium hydroxide | Potassium iodide | Potassium nitrate | Potassium permanganate | Potassium sulfate |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.0054 | 1.0072 | 1.0046 | 1.0053 | 1.0046 | 1.0068 | 1.0068 | 1.0056 | 1.0045 | 1.0051 | 1.0062 |
| 5 | 1.035 | 1.0437 | 1.0304 | 1.0342 | 1.031 | 1.0412 | 1.0419 | 1.036 | 1.0298 | 1.0322 | 1.0388 |
| 10 | 1.074 | 1.0904 | 1.0633 | 1.0703 | 1.065 | 1.068 | 1.0873 | 1.0762 | 1.0627 | 1.0806 | |
| 20 | 1.1601 | 1.1898 | 1.1328 | 1.1372 | 1.1818 | 1.1659 | 1.1326 | ||||
| 30 | 1.2593 | 1.2755 | 1.2813 | 1.2712 | |||||||
| 40 | 1.3746 | 1.4142 | 1.3881 | 1.3959 | |||||||
| 50 | 1.5404 | 1.5024 | |||||||||
| Density at 20°C, given as g/cm3 | |||||||||||
Conversion of the concentration from mass% to mol/kg (moles of solute/kg of water = molality):
For full table with Potassium Hydrogen Carbonate, Potassium Hydrogen Phosphate, Potassium Hydroxide, Potassium Iodide, Potassium Nitrate, Potassium Permanganate and Potassium Sulfate - rotate the screen!
| Mass% | Potassium bromide | Potassium carbonate | Potassium chloride | Potassium dihydrogen phosphate | Potassium hydrogen carbonate | Potassium hydrogen phosphate | Potassium hydroxide | Potassium iodide | Potassium nitrate | Potassium permanganate | Potassium sulfate |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.085 | 0.073 | 0.135 | 0.074 | 0.101 | 0.058 | 0.18 | 0.061 | 0.1 | 0.064 | 0.058 |
| 5 | 0.442 | 0.381 | 0.706 | 0.387 | 0.526 | 0.302 | 0.938 | 0.317 | 0.521 | 0.333 | 0.302 |
| 10 | 0.934 | 0.804 | 1.490 | 0.816 | 1.11 | 0.499 | 1.98 | 0.669 | 1.099 | 0.638 | |
| 20 | 2.101 | 1.809 | 3.353 | 2.497 | 4.456 | 1.506 | 2.473 | ||||
| 30 | 3.601 | 2.814 | 7.639 | 2.582 | |||||||
| 40 | 5.602 | 4.824 | 11.882 | 4.016 | |||||||
| 50 | 7.236 | 17.82 | |||||||||
| Molality at 20°C, given as mol/kg water | |||||||||||
Conversion of the concentration from mass% to mol/liter (moles of solute/liter of solution = molarity):
For full table with Potassium Hydrogen Carbonate, Potassium Hydrogen Phosphate, Potassium Hydroxide, Potassium Iodide, Potassium Nitrate, Potassium Permanganate and Potassium Sulfate - rotate the screen!
| Mass% | Potassium bromide | Potassium carbonate | Potassium chloride | Potassium dihydrogen phosphate | Potassium hydrogen carbonate | Potassium hydrogen phosphate | Potassium hydroxide | Potassium iodide | Potassium nitrate | Potassium permanganate | Potassium sulfate |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.084 | 0.073 | 0.135 | 0.074 | 0.1 | 0.058 | 0.179 | 0.061 | 0.099 | 0.064 | 0.058 |
| 5 | 0.435 | 0.378 | 0.691 | 0.380 | 0.515 | 0.299 | 0.929 | 0.312 | 0.509 | 0.327 | 0.298 |
| 10 | 0.903 | 0.789 | 1.426 | 0.786 | 1.064 | 0.491 | 1.938 | 0.648 | 1.051 | 0.62 | |
| 20 | 1.95 | 1.722 | 3.039 | 2.272 | 4.213 | 1.405 | 2.24 | ||||
| 28 | 3.175 | 2.584 | 6.851 | 2.297 | |||||||
| 40 | 4.62 | 4.03 | 9.896 | 3.364 | |||||||
| 50 | 5.573 | 13.389 | |||||||||
| Molarity at 20°C, given as mol/l solution | |||||||||||



